
Products and Services
Take2 Prophecy™ Test for NPCCancer Screening PackageStandard / Comprehensive Health Check PackagesTake2 Clarity™ Test for NPC MonitoringMembership ProgramFAQ
The Take2 Prophecy™ Test for Nasopharyngeal Cancer
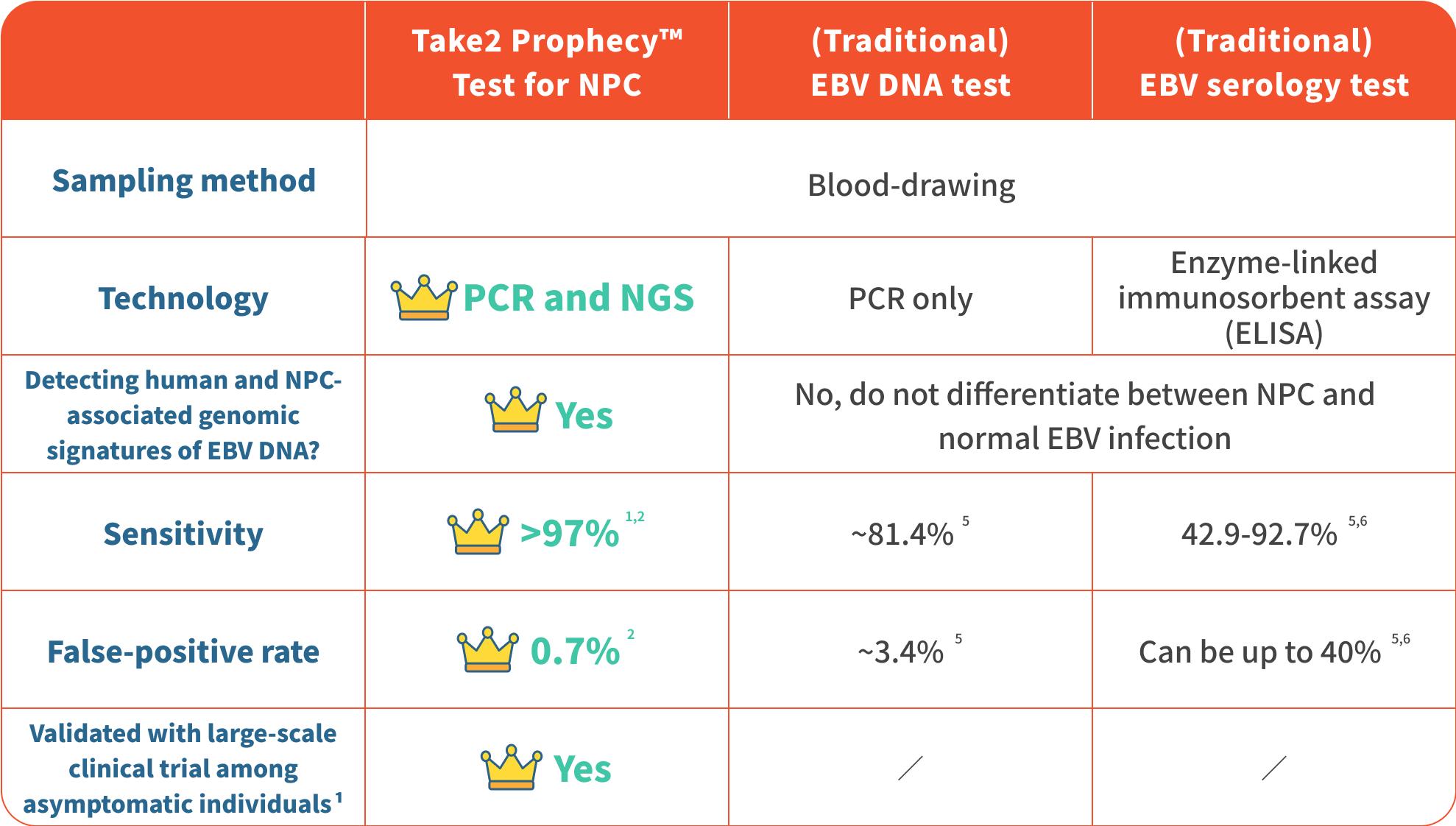
Take2 Prophecy™ Test for Nasopharyngeal Cancer (NPC) utilises Next-generation Sequencing (NGS) technology, that achieves > 97% sensitivity1,2 and 0.7% low false-positive rate2. Providing accurate and reliable results, the Take2 Prophecy™ Test for NPC ranks at the top over other similar tests. It only requires a blood draw to effectively identify early-stage NPC patients.
The Take2 Prophecy™ Test for NPC is suitable for individuals who intend to do routine check-ups, who are asymptomatic and with suspected symptoms.
Act Now and Stay Vigilant against NPC
NPC is one of the common cancers in HK3; it is the top cancer for men aged 20-444
Early NPC symptoms resemble flu and are difficult to distinguish (Some of the common symptoms include headaches, nasal blockage, tinnitus, etc.)
Perform regular NPC screening to catch the changes in your body before symptoms appear to allow timely treatment and interventions, thus increasing the chances to cure
Key Features of Take2 Prophecy™ Test for NPC:
Utilising Next-generation Sequencing technology
Achieving sensitivity > 97%1,2 with a remarkably low false positive rate (0.7%2)
Developed by local university leading research team
Being clinically proven by thousands of individuals1, the results have been published in international medical journals
Blood-drawing only, non-invasive, and minimal risk
Results will be ready soonest in 3 working days

Take2 Prophecy™ Test for NPC outperforms other similar tests:
NPC could be cured by detecting early! The survival rate of early-stage NPC patients could be higher than 90%1

Test Procedures
Doctor’s assessment
Collection of blood samples
Test performed at local laboratories
Test results ready in
3 - 7 working days#
Test results’ interpretation by the doctor
Test results’ interpretation by the doctor:
- Positive test result*Follow-up consultation with Ear, Nose & Throat Specialists* Positive: Human and NPC-associated genomic signatures of EBV DNA were detected
- Negative test result**Regular check-ups** Negative: Human and NPC-associated genomic signatures of EBV DNA were not detected
Important Note
Patients who have had organ transplant, currently have cancer or autoimmune diseases, or those currently receiving systemic glucocorticoid or immunosuppressive treatment are not recommended for early NPC detection using our test. If in doubt, please consult healthcare professionals for more information.
#From the day of receipt of samples, under normal circumstances, the analysis can be completed in about 3-7 working days
Patients who have had organ transplant, currently have cancer or autoimmune diseases, or those currently receiving systemic glucocorticoid or immunosuppressive treatment are not recommended for early NPC detection using our test. If in doubt, please consult healthcare professionals for more information.
#From the day of receipt of samples, under normal circumstances, the analysis can be completed in about 3-7 working days

Book our Service Now
Get tested to understand your risk of having NPC. Act now!
References
1.
Chan, K. C. Allen, et al. “Analysis of Plasma Epstein–Barr Virus DNA to Screen for Nasopharyngeal Cancer.”New England Journal of Medicine, vol. 377, no. 6, 2017, pp. 513–22.
2.
Lam, W. K. Jacky, et al. “Sequencing-Based Counting and Size Profiling of Plasma Epstein–Barr Virus DNA Enhance Population Screening of Nasopharyngeal Carcinoma.” Proceedings of the National Academy of Sciences, vol. 115, no. 22, 2018, pp. E5115–24.
3.
Hong Kong Cancer Registry. Hong Kong Hospital Authority, www3.ha.org.hk/cancereg/. Accessed 23 May 2021.
4.
Overview of Hong Kong Cancer Statistics of 2018. Hong Kong Hospital Authority, October 2020.
5.
Chang, Kai-Ping, et al. “Complementary Serum Test of Antibodies to Epstein-Barr Virus Nuclear Antigen-1 and Early Antigen: A Possible Alternative for Primary Screening of Nasopharyngeal Carcinoma.”Oral Oncology,vol. 44, no. 8, 2008, pp. 784–92.
6.
Tay, Joshua K., et al. “Screening in Nasopharyngeal Carcinoma: Current Strategies and Future Directions.”Current Otorhinolaryngology Reports, vol. 2, no. 1, 2013, pp. 1–7.
©2025 Take2 Health All Rights Reserved
Privacy PolicyTerms of UsePersonal Information Collection Statement繁/简/EN


